- Research
- Open access
- Published:
RETRACTED ARTICLE: Down-regulation of kappa opioid receptor promotes ESCC proliferation, invasion and metastasis via the PDK1-AKT signaling pathway
Cell Communication and Signaling volume 20, Article number: 35 (2022)
Abstract
Background
As a class of the opioid receptors, the kappa opioid receptor (KOR) has been verified to be a potential biomarker and therapeutic target for human malignant tumors. However, a thorough understanding of whether KOR affects progression of esophageal squamous cell carcinoma (ESCC) is still lacking. This study focused on exploring the effect of knocking down KOR in ESCC and its underlying mechanism.
Methods
Bioinformatics analysis was used to compare the different expression level of OPRK1 (KOR gene) in tumor and adjacent normal tissues, and predict the relationship between KOR expression and overall survival. RNA-sequence analysis was performed to detect the altered functions and mechanisms after down regulating KOR. The in vitro and in vivo assays were used to detect the effects of down-regulated KOR on cell proliferation, migration and invasion. Substrate gel zymography and 3D cell culture assays were used to find the effect of KOR knockdown on the degradation of extracellular matrix (ECM), and immunefluorescence was performed to detect the altered cytoskeleton. Western blotting and immunohistochemistry were used to explore the underlying mechanism pathway.
Results
Bioinformatics analysis revealed that the expression of OPRK1 was lower in tumor tissue than that in adjacent normal tissues, and lowered expression of KOR was associated with poorer overall survival. The in vitro assays demonstrated that down-regulation of KOR enhanced ESCC proliferation, metastasis and invasion. Western blotting revealed that down-regulation of KOR could activate PDK1-AKT signaling pathway, which actively regulated the cancer progression. Down-regulation of KOR enhanced the formation of invadopodia, secretion of matrix metalloproteinase-2 (MMP2) and rearrangement of cytoskeleton, which were positively related with the invasion of ESCC. KOR knockdown enhanced the tumor invasion and elevated the AKT phosphorylation in nude mice. The AKT kinase inhibition could reverse the effect of down-regulation of KOR.
Conclusion
KOR might act as a tumor suppressor in ESCC and down-regulation of KOR could enhance the ESCC tumor phenotype.
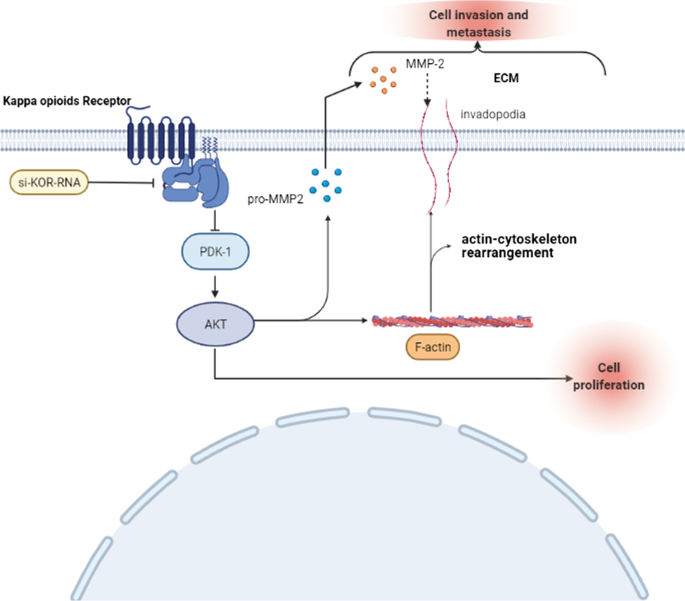
Graphical abstract
Video Abstract
Background
Esophageal cancer is the eighth most common type of cancer in the world [1]. The incidence of esophageal cancer has changed nearly 16 times globally, with the highest incidence in China, Central Asia, East Asia and South Africa [1, 2]. Among them, ESCC is the most important subtype of esophageal cancer [3]. Esophageal cancer has a poor prognosis and a high mortality rate. The characteristics and treatment of esophageal cancer appear to be changing. The disease is usually diagnosed late and the prospects for long-term survival are grim [1]. So far, the molecular mechanism of ESCC progression is unclear, leading to poor prognosis in ESCC patients [4]. Therefore, finding novel biomarkers and therapeutic targets is crucial to improve the diagnosis and treatment of ESCC.
Opioid receptors were first found in the central nervous system, and then in various organs and peripheral tissues [5, 6]. There are at least three types of opioid receptors, mu, delta, and kappa [5]. Opioid receptors have a variety of biological effects, including analgesia, cardiac protection, immunosuppression, respiratory depression, and gastrointestinal spasm [6,7,8,9,10]. In addition, opioid receptors have been found in a variety of primary human tumors and diverse of cancer cell lines [11]. Considerable evidence has been accumulated from both in vitro and in vivo studies, indicating that opioid receptors play a substantial role in regulating the growth of cancer cells. The expression of opioid receptors performs an important role in cell proliferation, invasion and metastasis [11,12,13,14].
KOR is a type of opioid receptors, the product of the OPRK1 gene. It has been reported that down-regulation of KOR is strongly associated with poor prognosis in hepatocellular carcinoma [15]. There are reports demonstrating that KOR expression regulates the proliferation of tumor cells, such as breast cancer and non-small cell lung cancer [16, 17]. The PI3K-Akt signaling pathway contributes to the enhanced proliferation following down-regulation of KOR in tumor cells [16, 17]. In addition, KOR is involved in the apoptosis of colorectal cancer cells via the β-catenin signaling axis [18]. KOR system also plays an important role in tumor angiogenesis [19]. Transplanted with Lewis lung cancer or B16 melanoma cells, KOR knockout mice significantly increased tumor angiogenesis and proliferation comparing with wild-type mice [20]. Activation of cAMP/PKA signaling pathway rescues the KOR-mediated inhibition of angiogenesis [21]. The involvement of KOR in carcinogenic events has considerable implications for understanding the etiology, diagnosis and treatment of neoplasia in humans. However, the possible effect of KOR on ESCC remains unknown. Thus, we designed a series of in vitro and in vivo experiments to explore the potential effects of KOR on ESCC.
Materials and methods
Cell lines and treatment
Human primary ESCC cell lines (KYSE30, 140, 150, 180 and 410) were cultured at 37 °C in a humidified atmosphere of 5% CO2 in RPMI 1640 medium (HYCLONE, USA) containing 10% fetal bovine serum. All cell lines were provided by Professor Li-Yan Xu (Institute of Oncologic Pathology, Medical College of Shantou University, Shantou 515041, PR China). Transiently-transfected cells were treated with HY-10249A (AKT kinase inhibitor, MCE, Weehawken, USA).
Bioinformatics analysis
The gene expression profile and clinical information were obtained from the Gene Expression Omnibus (GEO, www.ncbi.nlm.nih.gov/geo/). Based on the GSE23400 dataset, 53 adjacent normal tissue samples and 53 tumor tissue samples were selected for OPRK1 (KOR gene) expression analysis. A one-sided Student's t-test was used to analyze the OPRK1 expression values in the two groups. The mRNA expression data and corresponding clinical data of 179 ESCC patients were obtained from the GSE53625. We used this dataset to analyze the relationship between KOR expression level and the survival of ESCC patients. Differences were considered statistically significant when P < 0.05.
Gene ontology (GO) and kyoto encyclopedia of genes and genomes (KEGG) enrichment analysis
Appropriate amount of TRIzol lysate was added into the cell sample tubes (the reference dosage was 1 ml TRIzol per 1 × 106 cells), after vortex mixing, the cells were placed at room temperature for 5 min to fully lysis. Oligo (dT) magnetic beads were used to enrich the mRNA and interrupt reagents were added to fragment the mRNA. The synthesized cDNA chain was added to the interrupted mRNA for synthesis of two-stranded cDNA. PCR reaction system was prepared and reaction procedure was set up to amplify the linker products. After denaturing the PCR product into a single chain, the cyclization reaction system was prepared to obtain the final library. The single-stranded circular DNA molecule was replicated by rolling ring and high-density DNA nanochip technology combined with probe anchoring polymerization (cPAS) was used to obtain 50 bp/100 bp/150 bp sequencing read length. The differentially-sequencing was selected and subjected to GO and KEGG pathway analyses. GO analysis (http://geneontology.org/) describes and classifies related genes according to biological processes, cell components, and molecular functions. For KEGG analysis (http://www.genome.jp/kegg/), the differentially-affected pathways were sorted according to their enrichment scores.
Cell transfection
Small interfering RNA against KOR (si-KOR) and negative control si-RNA (NC) were synthesized by GenePharma (Suzhou, China). The sequencing of si-KOR-1 was 5′-CAGCAACCAACAUUUACAUTT-3′, the si-KOR-2 was 5′-CGCCUUAGGCUA-UACCAACTT-3′ and the si-KOR-3 was 5′-CACUAGCAGAGUCCGAAAUTT-3′. Si-KOR-2 was proved effective in knocking down and was applied to subsequent rescue experiment. For stably knocking down KOR, plasmid containing short hairpin RNA against KOR (sh-KOR) and corresponding negative control empty plasmid (NC) were synthesized and purchased from GENECHEM Company (Shanghai, China). The sequencing of sh-KOR-1 was gcACTAGCAGAGTCCGAAATA, the sh-KOR-2 was gaAGGCAAAGATCATCAATAT and the sh-KOR-3 was cgATGTCATTGAGTGC-TCCTT. Sh-KOR-2 was proved effective in knocking down and was applied to subsequent mice experiment. Transfections were performed with the Lipofectamine 3000 Transfection Reagent (Thermo, USA). Cells were harvested for further study at 24 h post-transfection. Transfected cells were treated with puromycin to establish cells with stable knockdown of KOR.
Cell migration and invasion assays
Migration and invasion assays were performed using transwell chambers. For invasion assays, 1 × 105 cells were seeded onto the top Matrigel-coated chamber with 8-μm pores (BD Falcon, USA), and the bottom chamber was filled with medium with 10% fetal bovine serum (for the migration assay, cells were directly plated on an uncoated chamber). After 24 h (migration assays) or 48 h (invasion assays), we removed the top layer cells in chambers and the membranes were fixed and stained with hematoxylin (Baso, China). The cell numbers were quantified by counting 10 random fields under microscope (200x, Olympus IX73, Japan).
Wound healing assay
Cells were seeded into six-well dish, cultured in each complete medium, and grown to confluence overnight. The cells were scratched using a standard 200 μl tip, and the debris was removed by washing the cells with serum-free medium. Subsequently, the scratched cells were cultured with 2% fetal bovine serum medium and serial photographs were obtained at different time points using microscope (200x, Olympus IX73, Japan).
CCK-8 assay
A Cell Counting Kit-8(CCK-8) assay was conducted to assess the cell proliferation. Initially, cells were seeded into the 96 wells plates and incubated at 37 °C overnight. Afterwards, Cell Counting kit-8 solution (CCK-8, Dojindo, Mashikimachi, Japan) was added to the cells, which were further incubated for 1 h. Absorbance at 450 nm was measured at 0, 24, 48, 72 and 96 h using a plate microplate reader (Multiskan FC, Thermo, USA).
Colony formation assays
The cells were seeded into 6-well plates at 2 × 103 per well and incubated for 2 weeks for the colony formation assay. Cells were then washed twice with PBS, fixed with methanol/acetic acid (3:1, v/v), and stained with 0.5% crystal violet (Sigma, China). Photography of colonies was performed using a ChemiDoc XRS + Imaging System (Bio-Rad, USA). The number of colonies was counted with the Image J.
Substrate gel zymography
The gelatin substrate zymogram was performed in a 10% prefabricated SDS-PAGE gel with 1 mg / ml gelatin (Thermo, USA). Equal amounts of proteins were diluted into 2 × Tris–glycine SDS sample buffer and electrophoretically separated under non-reducing conditions. The protein was renatured for 30 min in renaturation buffer (Thermo, USA), and then, the gel was incubated in the development buffer (Thermo, USA) for 30 min and in the same buffer at 37 °C overnight. The gel was stained with Simply Blue SafeStain (Thermo, USA), and clear bands of gelatinase activity were visible on the blue background.
3D cell culture
Three-dimensional (3D) culture was performed using Matrigel basement membrane matrix (Corning, USA). KYSE30 or KYSE180 cells were mixed with Matrigel matrix at 8 × 105 cells/100ul, which were seeded into 6-well plates and cultured in RPMI 1640 medium containing 10% fetal bovine serum. Cells were incubated at 37ºC, under 5% CO2-95% air and observed every 3 h to monitor the formation of invadopodia using microscope (200x, Olympus IX73, Japan).
Immunofluorescence
Cells were grown on coverslips in 24-well plates. When cells reached 80–90% confluence, medium was aspirated and cells were washed with 500 μl PBS. Cells were fixed in 4% formaldehyde for 10 min and washed as described above. The cells were permeabilized by 0.1% Triton X-100 in PBS for 3 min at room temperature. After washing with PBS, the cells were blocked for 1 h in PBS containing 5% donkey serum.Then, 30 µl anti-KOR antibody (1:100, Abcam, UK) was added to each coverslip, which were incubated overnight at 4 °C in the dark. After washing, coverslips were stained with Acti-stain 488 phalloidin (Cytoskeleton, USA, #PHDG1) and Dylight 594-conjugated AffiniPure donkey anti-rabbit IgG (H + L) (Jackson, #99212) for 1 h. After staining with DAPI for 1 h, the coverslips were mounted with antifade mounting medium (Beyotime, China, #P0126) on slides, and cells were observed with a confocal microscope (LSM 800, ZEISS, Germany).
Western blotting
Cell lysates were prepared by mixing RIPA lysis buffer (RIPA; Pierce, Rockford, IL) with Protease and Phosphatase Inhibitor Cocktail (78445, EDTA-free, 100 ×, Thermo, USA). Protein concentration was determined by a bicinchoninic acid kit (Pierce Biotechnology, USA). The proteins were then separated by electrophoresis on a 10% sodium lauryl sulfate–polyacrylamide gel, and then transferred to a polyvinylidene difluoride membrane (Roche, Switzerland). The membrane was blocked with Tris-buffered saline containing 5% skimmed milk for one hour at room temperature. Thereafter, the membranes were incubated with primary antibodies against KOR (1:1000, Abcam, UK), glyceraldehyde-3phosphate dehydrogenase (GAPDH, 1:5000, Thermo, USA), protein kinase B (AKT, 1:1000, CST, USA), phosphorylated AKT Ser473 (pAKT(S473), 1:1000, CST, USA), 3-phosphoinositide-dependent protein kinase-1 (PDK1, 1:1000, CST, USA) and phosphorylated PDK1 (pPDK1, 1:1000, CST, USA) at 4 °C overnight. Subsequently, membranes were incubated with HRP-conjugated secondary antibody goat anti-rabbit IgG (1:5000, Thermo, USA) or goat anti-mouse IgG (1:5000, Thermo, USA) at room temperature for 2 h. GAPDH was used as the internal control. Band signals were detected using SuperSignal West Pico PLUS Chemiluminescent Substrate (Thermo, USA). Photography of the band signals was performed using a ChemiDoc XRS + Imaging System (Bio-Rad, USA).
Tumor invasion assay in vivo
All animal experiments complied with the policy of Shantou University Medical College on the care and use of laboratory animals. Four-week-old male NU/NU nude mice were obtained (Charles River, China) and bred under specific pathogen-free conditions. KYSE30 cells stably knock down for KOR, or control cells, were subcutaneously injected into the foot pad of the mice (5 mice per group). Over a period of 5 weeks, tumor invasion in the mice was observed by measuring the volume of proximal invasive tumor. Upon termination of the experiment, mice were euthanized and tumors were excised.
Immunohistochemistry
Tissues were fixed in formalin and embedded in paraffin for immunohistochemistry. In short, tissue sections are dewaxed in xylene and then rehydrated with a graded series of ethanol. In order to inhibit endogenous peroxidase activity, the sections were immersed in 0.3% peroxidase-methanol for 30 min. During the antigen extraction process, the slices were pretreated with citrate buffer in a microwave oven at 100 °C for 15 min. Sections were incubated with primary antibody against KOR (Abcam, UK), pAKT(S473, CST, USA) at 4 °C overnight at a dilution of 1:100. After consultation with the manufacturer, the Ultra Vision Quanto Inspection System HRP DAB Kit (Thermo, USA) was used to visualize the sections. The stained sections were counter-stained with hematoxylin, and micrographs were taken with an Olympus BX53 microscope (Japan).
Statistical analysis
In this study, statistical analyses were performed with SPSS 17.0 and GraphPad Prism 7.0 software. All experiments were performed with at least three independent replicates. Data were represented as mean ± standard deviation. Differences were considered statistically significant at P < 0.05 (*), P < 0.01 (**), P < 0.001 (***) and P < 0.0001 (****). The one/two-sided Student's t-test were separately used to compare the statistical differences.
Results
KOR expression might be related with ESCC progression
The microarray data (GSE23400) was used to analyze the different expression level of OPRK1 (KOR gene) between the tumor and adjacent normal tissues. We screened 53 adjacent normal tissue samples and 53 tumor tissue samples for OPRK1 expression analysis. We found that OPRK1 expression in adjacent normal tissue was significantly higher than that in tumor tissue (P < 0.05; Fig. 1A). In addition, we used the GSE53625 to analyze the relationship between KOR expression and the survival of ESCC patients. Kaplan–Meier survival curves indicated that the patients with low KOR expression had a significantly lower overall survival than patients with high KOR expression (P < 0.05; Fig. 1B). To study the expression of KOR in a panel of five ESCC cell lines, we initially examined KOR protein level in ESCC cell lines (Fig. 1C). High KOR protein levels were observed in KYSE30 and KYSE180 cells, while low expression in KYSE150 and KYSE410 cells. We detected the proliferation and invasion characteristics of four cell lines mentioned above. The results showed that KYSE150 and KYSE410 cells had more active characteristics of proliferation and invasion than KYSE30 and KYSE180 cells. (Fig. 1 D-E).
KOR expression might be related with ESCC progression. A Different expression level of OPRK1 (KOR gene) in tumor and adjacent normal tissue. The expression level of OPRK1 in adjacent normal tissue was higher than that of tumor tissue (P = 0.04). B Kaplan–Meier survival curves of patients classified into high- and low-expression KOR groups. Compared with the high KOR expression group, ESCC patients with low KOR expression had a lower survival time (P = 0.03). C Relative expression of KOR was detected in five ESCC cell lines by western blotting and KOR expression was normalized to GAPDH. D–E KYSE150 and KYSE410 cells showed more active proliferation and invasion characteristics than KYSE30 and KYSE180 cells
KOR downregulation promotes ESCC cell proliferation, invasion and migration
KOR was highly expressed in KYSE30 and KYSE180 cells (Fig. 1C). We knocked down KOR in these two cell lines for subsequent experiments (Fig. 2A). To explore the potential variation of function, GO analysis was performed to predict the altered biological processes. The GO terms were determined by calculating the enrichment score. The top twenty highest and most significant GO terms were shown in Fig. 2B. The red framed GO terms in Figure 2B had significant differences, including cell adhesion and cell proliferation, which were demonstrated in subsequent experiments.
KOR knockdown promotes ESCC cell proliferation, invasion and migration. A KOR protein levels in KYSE30 and KYSE180 decreased after transfection with the small interfering RNA against KOR (si-KOR-1, 2), as comparing with the negative control si-RNA (NC). B Gene Ontology (GO) analysis of dysregulated genes in KOR knockdown cells comparing with control. C–D Down-regulation of KOR enhanced the proliferation of KYSE30 and KYSE180 cells. E Down-regulation of KOR enhanced the invasive ability of KYSE30 and KYSE180 cells. F–G Down-regulation of KOR enhanced the migration of KYSE30 and KYSE180 cells
Firstly, we determined whether KOR could regulate proliferation of ESCC cells. Results of colony formation and CCK-8 assays suggested that knocking down KOR led to enhance cell proliferation comparing with control (Fig. 2C, D). Furthermore, cell invasion assays demonstrated that down-regulation of KOR effectively enhanced the invasive ability of KYSE30 and KYSE180 cells (Fig. 2E). Cell migration assays including transwell migration and wound healing assays revealed that down-regulation of KOR effectively promoted the migratory ability of KYSE30 and KYSE180 cells (Fig. 2F, G).
KOR downregulation activates pPDK1-mediated-pAKT(S473) phosphorylation
To examine the impact of knocking down KOR on signaling pathways, pathway analysis was performed using the KEGG database, which identified twenty pathways with significant differences in up-regulated gene expression (Fig. 3A). Three pathways, highlighted with red boxes in Fig. 3A, were significantly upregulated, and included the PI3K-Akt signaling pathway, ECM-receptor interaction and regulation of actin cytoskeleton. As shown in the western blot, the pAKT (S473) level was elevated in KOR knockdown cells than control, while AKT levels did not change (Fig. 3B). Subsequently, we detected the signaling activity upstream of AKT and found that the pPDK1 level was elevated, indicating activation of PDK1, in the absence of changes in PDK1 levels following knock down of KOR (Fig. 3B).
KOR downregulation activates pPDK1-mediated-pAKT(S473) phosphorylation. A KEGG analysis of pathways enriched for genes up-regulated following KOR knockdown. The results showed that these genes were involved in the PI3K-Akt pathway, ECM-receptor interaction and regulation of actin cytoskeleton. B KOR knockdown elevated the phosphorylation level of pPDK1 and pAKT (S473), in ESCC cells, in the absence of changes in total protein expression of PDK1/AKT
KOR downregulation enhances ECM degradation and mediates actin cytoskeletal rearrangement
We conducted zymography analyses to investigate the key enzymes involved in degrading ECM, and found that the activity of MMP-2 in KOR knockdown cells was clearly elevated (Fig. 4A). We therefore examined the role of KOR in invadopodia formation by using 3D cell culture assay. The number of invadopodia in KOR knockdown cells was increased, as comparing with control (Fig. 4B). The above results suggested that KOR inhibition enhanced tumor cell invasion and metastasis through degradation of the ECM. At the same time, by immunofluorescence, we found that the stress fibers in KOR knockdown cells were more disordered than control, with the F-actin cytoskeleton radially rearranged in the periphery of KOR knockdown cells (Fig. 4C).
KOR downregulation enhances ECM degradation and mediates actin cytoskeletal rearrangement. A Substrate gel zymography showed that KOR knockdown induced MMP2 secretion. B 3D cell culture indicated that KOR knockdown enhanced invadopodia information. C Immunofluorescence imaging showed that down-regulated KOR mediated actin cytoskeleton rearrangement, especially in extended filopodia
Decreased KOR promotes invasion in vivo
Based on the above results, we further explored whether the expression of KOR could regulate invasion in vivo. Western blotting demonstrated that KYSE30 cells transfected with sh-KOR-2 stably expressed low levels of KOR (Fig. 5A). Subsequently, we injected the sh-KOR and NC cells into right foot pad of nude mice, and observed the tumor growth and invasion (Fig. 5B). We excised tumor tissues from the mice after five weeks and separated the primary and proximal tumor tissue (Fig. 5C). KOR knockdown tumors were larger and showed greater proximal invasion, as comparing with control. Immunohistochemistry assays showed that tumor tissues of the sh-KOR group exhibited weaker staining of KOR and stronger staining of pAKT (S473) than the NC group (Fig. 5D).
Decreased KOR promotes invasion in vivo. A Protein expression of KOR in KYSE30 cells stably transfected with the plasmid containing shRNA against KOR (sh-KOR), comparing with the empty plasmid (NC). B Tumor formation in nude mice. Sh-KOR cells or NC cells were injected into the right foot pad of nude mice, and tumor volumes were measured after 5 weeks. C Surgically removing tumor tissues from nude mice 5 weeks post-inoculation, and separating the primary and proximal invasive tumors. D Histological images showed that tissues of the sh-KOR tissue exhibited weaker staining of KOR, while stronger staining of pAKT than the NC
AKT kinase inhibition reverses the effects of KOR knockdown on proliferation, invasion and migration
In our study, we designed a series of recue experiments to verify that AKT activation was responsible for the proliferation, invasion and migration following KOR knockdown. We detected pAKT(S473) levels after treatment with the AKT kinase inhibitor HY-10249A (0.5 μM) (Fig. 6A). The AKT kinase inhibitor inhibited the phosphorylation of pAKT(S473), which attenuated the proliferative ability, reversing the growth-enhancing effects of KOR knockdown (Figs. 6B, C). Similarly, we found that the inhibition of pAKT(S473) suppressed cell invasion (Fig. 6D) and cell migration in transwell and wound healing assays (Figs. 6E, F).
The AKT kinase inhibitor (HY-10249A) reverses KOR knockdown-mediated enhancement of proliferation, invasion and migration. A Levels of pAKT (S473) in KYSE30 and KYSE180 cells were decreased after treatment with HY-10249A (0.5 μM). B, C HY-10249A inhibited the proliferation of KYSE30 and KYSE180 cells, as reversing the effect of KOR knockdown. D. HY-10249A inhibited the invasive ability of KYSE30 and KYSE180 cells, as reversing the effect of KOR knockdown. E–F HY-10249A inhibited the migratory ability of KYSE30 and KYSE180 cells, as reversing the effect of KOR knockdown
AKT kinase inhibition reverses the effect of KOR knockdown on ECM degradation and actin cytoskeletal rearrangement
Based on above results, we proposed that AKT activation could enhance ESCC invasion and metastasis by inducing the secretion of MMP2 and rearranging F-actin cytoskeleton. Therefore, we investigated whether the HY-10249A (0.5 μM) could reverse the mechanisms of KOR knockdown on invasion and migration. Inhibition of pAKT(S473) reversed the induced MMP2 secretion following KOR knockdown (Fig. 7A). In addition, we found that the inhibition of pAKT(S473) could further inhibit the invadopodia formation in 3D cell culture, as reversing the effect of KOR knockdown (Fig. 7B). Similarly, pAKT(S473) inhibition could reverse the effect of down-regulated KOR on the disordered stress fibers and rearranged F-actin cytoskeleton (Fig. 7C).
The AKT kinase inhibitor (HY-10249A) reverses the effect of KOR knockdown on ECM degradation and actin cytoskeletal rearrangement. A. HY-10249A reversed the effect of KOR knockdown on inducing MMP2 secretion. B. HY-10249A reversed the effect of KOR knockdown on invadopodia formation. C. HY-10249A reversed the effect of KOR knockdown on actin cytoskeletal rearrangement
Discussion
It had been reported that KOR could regulate proliferation, migration and invasion of tumor [15,16,17,18,19,20,21]. Bioinformatics analysis indicated that the expression of KOR might be associated with ESCC progression. Our results showed that down-regulation of KOR enhanced ESCC proliferation, invasion and metastasis both in vitro and in vivo. Consistent with this, we demonstrated that down-regulation of KOR enhanced ESCC proliferation, invasion and metastasis via PDK1-AKT signaling pathway.
According to the literature, esophageal cancer cell lines (KYSE series) were established from the resected specimens of patients with esophageal cancer. KYSE30 and 180 cells were originated from well-differentiated squamous cell carcinoma and KYSE150 and 410 cells were originated from the poor [22]. Our invasion and colony assays also seemed to prove that the invasion and proliferation characteristic of KYSE30 and 180 cells were less active than KYSE150 and 410 cells (Fig. 1D). Combined with bioinformatics analysis (Fig. 1A, B), we speculated that KOR might be related with ESCC progression, acting as a tumor suppressor. KOR was initially identified as a pain-related receptor, and its genetic polymorphisms were found to be related with substance dependence [23]. 36G > T single nucleotide polymorphism (SNP) on KOR gene had been reported to be associated with opioid dependence [24]. But as far as we known, there are no relevant statistical evidence focused on the relationship between KOR gene polymorphisms and ESCC progression, which might be a new research direction.
Migration and invasion are usually accompanied by degradation of the ECM and cytoskeletal remodeling. Usually, cytoskeletal disorders were related to the invasion and metastasis of tumor cells [25]. In terms of mechanism, invadopodia were invasive micro-scale structures that degraded ECM through a combination of membrane binding and soluble matrix metalloproteinase (MMPs) [26]. The actin cytoskeleton played an important role in the cell movement, differentiation, division and membranes organization. These processes required the coordinated renewal and reconstruction of actin fibers [27]. At the same time, ECM-receptor interaction and regulation of the actin cytoskeleton experienced major changes following KOR knockdown, as identified by KEGG analysis. Consistent with this, we demonstrated that ECM degradation and rearrangement of the actin cytoskeleton contributed to ESCC invasion and metastasis following down-regulation of KOR.
There is increasing evidence that PDK1 is an interesting and untapped target in cancer treatment. PDK1 was related to signaling pathways that were frequently changed in cancer, such as PI3K/AKT, Ras/MAPK and Myc pathways. In recent years, PDK1-mediated AKT activation had been extensively studied [28]. AKT was overexpressed in many human tumors, and promoted cancer cell growth, metabolism and survival. The PDK1-AKT pathway regulated the proliferation, invasion and migration of tumors [28,29,30,31,32]. Previous studies demonstrated that KOR could play a role in cancer progression via the AKT signaling pathway [16, 17]. Based on our rescue experiments, we showed that down-regulated KOR enhanced ESCC proliferation via AKT activation. In addition, there were studies showing that AKT also regulated the secretion of MMPs and rearrangement of the actin cytoskeleton [33, 34]. Our rescue experiments also demonstrated that down-regulation of KOR promoted ECM degradation and actin cytoskeletal rearrangement via elevating AKT phosphorylation so as to promote the invasion and metastasis of ESCC. The results of our study were presented as a graphic abstract to illustrate how KOR knockdown regulated ESCC progression.
Conclusions
Our findings show that down-regulation of KOR can directly activate ESCC proliferation, invasion and metastasis via a PDK1-AKT signaling pathway, indicating that KOR might serve as a tumor suppressor and therapeutic target for ESCC. To our knowledge, this is the first report demonstrating that KOR activity inhibits ESCC. Opioids are usually opioid receptor agonists and widely used as effective analgesics in clinical practice. It had also been reported that opioids were associated with tumor recurrence-free survival [35]. However, it is still unclear whether KOR nature ligands take part in the regulation of ESCC, and future studies are warranted in this direction. A better understanding of the effects on cancer by KOR system should greatly contribute to potential therapeutic strategies for cancers beyond the relief of cancer pain.
Availability of data and materials
The GEO database (https://www.ncbi.nlm.nih.gov/geo/) were used for detecting expression of OPRK1 and survival analyses.
Change history
27 June 2023
This article has been retracted. Please see the Retraction Notice for more detail: https://doi.org/10.1186/s12964-023-01205-1
Abbreviations
- KOR:
-
Kappa opioid receptor
- ESCC:
-
Esophageal squamous cell carcinoma
- GEO:
-
Gene Expression Omnibus
- ECM:
-
Extracellular matrix
- GO:
-
Gene Ontology
- KEGG:
-
Kyoto Encyclopedia of Genes and Genomes
- si-KOR:
-
Small interfering RNA against KOR
- sh-KOR:
-
Short hairpin RNA against KOR
- NC:
-
Negative control
- 3D:
-
Three-dimensional
- CCK-8:
-
Cell Counting Kit-8
- GAPDH:
-
Glyceraldehyde-3phosphate dehydrogenase
- AKT:
-
Protein kinase B
- pAKT:
-
Phosphor-AKT
- PDK1:
-
3-Phosphoinositide-dependent protein kinase-1
- pPDK1:
-
Phosphor-PDK1
- MMP-2:
-
Matrix metalloproteinase-2
- MMP:
-
Matrix metalloproteinase
- SNP:
-
Single nucleotide polymorphism
References
Huang FL, Yu SJ. Esophageal cancer: risk factors, genetic association, and treatment. Asian J Surg. 2018;41(3):210–5.
Torre LA, Siegel RL, Ward EM, et al. Global cancer incidence and mortality rates and trends–an update. Cancer Epidemiol Biomarkers Prev. 2016;25(1):16–27.
Codipilly DC, Qin Y, Dawsey SM, et al. Screening for esophageal squamous cell carcinoma: recent advances. Gastrointest Endosc. 2018;88(3):413–26.
Talukdar FR, di Pietro M, Secrier M, et al. Molecular landscape of esophageal cancer: implications for early detection and personalized therapy. Ann N Y Acad Sci. 2018;1434(1):342–59.
Minami M, Satoh M. Molecular biology of the opioid receptors: structures, functions and distributions. Neurosci Res. 1995;23:121–45.
Bodnar RJ. Endogenous Opiates and Behavior: 2018. Peptides. 2020; 132:170348.
Guo W, Imai S, Yang JL, et al. NF-KappaB pathway is involved in bone marrow stromal cell-produced pain relief. Front Integr Neurosci. 2018;12:49.
See Hoe L, Patel HH, Peart JN. Delta opioid receptors and cardioprotection. Handb Exp Pharmacol. 2018;247:301–34.
He C, Li H, Zhang J, et al. In vitro and in vivo characterization of the bifunctional μ- and δ- opioid receptors ligand MCRT on mouse gastrointestinal motility. Neuropeptides. 2019;74:82–7.
Ryu JH, Do SH, Han SH, et al. Morphine reduces mouse microglial engulfment induced by lipopolysaccharide and interferon-γ via δ opioid receptor and p38 mitogen-activated protein kinase. Neurol Res. 2018;40(7):600–6.
Fichna J, Janecka A. Opioid peptides in cancer. Cancer Metastasis Rev. 2004;23:351–66.
Wu Q, Chen X, Wang J, et al. Nalmefene attenuates malignant potential in colorectal cancer cell via inhibition of opioid receptor. Acta Biochim Biophys Sin (Shanghai). 2018;50(2):156–63.
Zhang Y, Wei Y, Li X, et al. microRNA-874 suppresses tumor proliferation and metastasis in hepatocellular carcinoma by targeting the DOR/EGFR/ERK pathway. Cell Death Dis. 2018;9(2):130.
Qu N, Wang X, Meng Y, Shan F. Prospective oncotarget for gynecological cancer: Opioid growth factor (OGF)—opioid growth factor receptor (OGFr) axis. Int Immunopharmacol. 2019;75:105723.
Chen D, Chen Y, Yan Y, et al. Down-regulation of the tumour suppressor κ-opioid receptor predicts poor prognosis in hepatocellular carcinoma patients. BMC Cancer. 2017;17(1):553.
Kharmate G, Rajput PS, Lin YC, et al. Inhibition of tumor promoting signals by activation of SSTR2 and opioid receptors in human breast cancer cells. Cancer Cell Int. 2013;13(1):93.
Kuzumaki N, Suzuki A, Narita M, et al. Effect of κ-opioid receptor agonist on the growth of non-small cell lung cancer (NSCLC) cells. Br J Cancer. 2012;106(6):1148–52.
Wang H, Wen C, Chen S, et al. Toosendanin-induced apoptosis in colorectal cancer cells is associated with the κ-opioid receptor/β-catenin signaling axis. Biochem Pharmacol. 2020;177:114014.
Yamamizu K, Hamada Y, Narita M. κ Opioid receptor ligands regulate angiogenesis in development and in tumours. Br J Pharmacol. 2015;172(2):268–76.
Yamamizu K, Furuta S, Hamada Y, et al. к Opioids inhibit tumor angiogenesis by suppressing VEGF signaling. Sci Rep. 2013;3:3213.
Yamamizu K, Furuta S, Katayama S, et al. The κ opioid system regulates endothelial cell differentiation and pathfinding in vascular development. Blood. 2011;118(3):775–85.
Shimada Y, Imamura M, Wagata T, Yamaguchi N, Tobe T. Characterization of twenty-one newly established esophageal cancer cell lines. Cancer. 1991;69:277–84.
Yuferov V, Fussell D, LaForge KS, et al. Redefinition of the human kappa opioid receptor gene (OPRK1) structure and association of haplotypes with opiate addiction. Pharmacogenetics. 2004;14(12):793–804.
Gerra G, Leonardi C, Cortese E, et al. Human kappa opioid receptor gene (OPRK1) polymorphism is associated with opiate addiction. Am J Med Genet B Neuropsychiatr Genet. 2007;144B(6):771–5.
Peng JM, Bera R, Chiou CY, et al. Actin cytoskeleton remodeling drives epithelial-mesenchymal transition for hepatoma invasion and metastasis in mice. Hepatology. 2018;67(6):2226–43.
Kumar S, Das A, Barai A, et al. MMP secretion rate and inter-invadopodia spacing collectively govern cancer invasiveness. Biophys J. 2018;114(3):650–62.
Li X, Wang J. Mechanical tumor microenvironment and transduction: cytoskeleton mediates cancer cell invasion and metastasis. Int J Biol Sci. 2020;16(12):2014–28.
Gagliardi PA, Puliafito A, Primo L. PDK1: at the crossroad of cancer signaling pathways. Semin Cancer Biol. 2018;48:27–35.
Emmanouilidi A, Falasca M. Targeting PDK1 for chemosensitization of cancer cells. Cancers (Basel). 2017;9(10):140.
Revathidevi S, Munirajan AK. Akt in cancer: mediator and more. Semin Cancer Biol. 2019;59:80–91.
Pan W, Li W, Zhao J, et al. lncRNA-PDPK2P promotes hepatocellular carcinoma progression through the PDK1/AKT/Caspase 3 pathway. Mol Oncol. 2019;13(10):2246–58.
Bamodu OA, Chang HL, Ong JR, et al. Elevated PDK1 expression drives PI3K/AKT/MTOR signaling promotes radiation-resistant and dedifferentiated phenotype of hepatocellular carcinoma. Cells. 2020;9(3):746.
Zhu Y, Yan L, Zhu W, et al. MMP2/3 promote the growth and migration of laryngeal squamous cell carcinoma via PI3K/Akt-NF-κB-mediated epithelial-mesenchymal transformation. J Cell Physiol. 2019.
Cenni V, Sirri A, Riccio M, et al. Targeting of the Akt/PKB kinase to the actin skeleton. Cell Mol Life Sci. 2003;60(12):2710–20.
Montagna G, Gupta HV, Hannum M, et al. Intraoperative opioids are associated with improved recurrence-free survival in triple-negative breast cancer. Br J Anaesth. 2021;126(2):367–76.
Acknowledgements
We thank Professor Li-Yan Xu (Institute of Oncologic Pathology, Medical College of Shantou University, Shantou 515041, PR China) for providing ESCC cell lines.
Funding
This work was supported by grants from the Medical Research Foundation of Guangdong Province (No. A2015026), the Natural Science Foundation of Guangdong Province (No. 2018A030313069 and No. 2016A030313069), and the Doctoral Foundation of Shantou University Medical College (No. 413796).
Author information
Authors and Affiliations
Contributions
Huang HM contributed to cell culture, invasion assay, migration assay, proliferation assay, mice assay, data analysis and interpretation and drafting; He XH contributed to immunohistochemistry assay, invasion assay, proliferation assay, data collection and date analysis; Huang XY contributed to study design, data collection and revision of the manuscript; Wang GY contributed to bioinformatics analysis; Xia QX contributed to survival analysis; Zhang YF and Du ZP contributed to study design, data analysis and interpretation, and revision of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
No potential competing interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article has been retracted. Please see the retraction notice for more detail: https://doi.org/10.1186/s12964-023-01205-1
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Huang, HM., He, XH., Huang, XY. et al. RETRACTED ARTICLE: Down-regulation of kappa opioid receptor promotes ESCC proliferation, invasion and metastasis via the PDK1-AKT signaling pathway. Cell Commun Signal 20, 35 (2022). https://doi.org/10.1186/s12964-022-00833-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12964-022-00833-3









